Marie is the first woman to win a Nobel Prize.

Did considerable work in the investigations of radioactivity. From this experiment he postulated that atoms are formed of a small dense positively charged nucleus "orbited" by negatively charged electrons. He discovered that the particles bounced off of something dense in the foil. In a classic experiment he bombarded a thin sheet of gold foil with alpha particles (He nuclei: 2 protons + 2 neutrons). He measured the static electrical charge on microscopic oil droplets by balancing droplets between charged plates.Įrnest Rutherford. He calculated the charge on the electron with his famous oil drop experiment. He calculated the charge to mass ratio e/m for the electron by careful observations of the curvature of an electron beam in cathode ray tubes in a magnetic field. He postulated a "Plum Pudding" model for atoms.
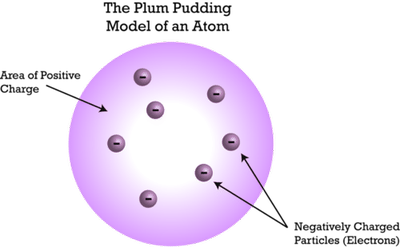
He showed that atoms could be further subdivided into negative (which he named electrons) and positive components.
#JJ THOMSON CATHODE RAY EXPERIMENT FOUND THE NUCLEUS TV#
Studied the newly invented cathode ray tube (a TV screen is a cathode ray tube). Einstein analyzed and explained the phenomenon in 1905. It is caused by the molecular movement of the molecules of the fluid in which the particle is suspended. It was discovered by the biologist Robert Brown in 1827. The jerking movement of microscopic particles only visible under a high powered microscope. The elements arranged in families, ordered in order of increasing atomic number (the number of protons in the nucleus).īrownian Motion. Discovered a periodicity in the properties of elements. Gases (under ideal conditions) combine to form compounds in the same ratios by volume equal volumes of gases under the same conditions of temperature and pressure contain the same number of particles. Formalized the discovery of Lavoisier into the "Law of Definite Proportions:" When atoms combine to form a particular compound, they always combine in the same ratios by weight.Īvogadro's law. In this case the ratio by weight of Hg to O was always 12.5 to 1.ĭalton. By heating mercuric oxide (HgO) and allowing it to decompose into its component elements he discovered that the component elements mercury and the gas (oxygen) given off always combine in the same ratios by weight to form the compound. Preformed the classic experiment with mercuric oxide.

Ancient Greek philosopher who advocated the atomic theory of matter. Matter could be subdivided into these fundamental units and no further.ĭemocritus. Atomic Theory: Substances and elements are made up of small units called atoms.

